 
Balance the given equation for the decomposition of calcium nitrate. Let us balance an equation for some practice. Example for Balancing a Chemical Equation After balancing the chemical equation, make sure that the total number of atoms of an element present on the reactant and product side must be equal. The stoichiometric coefficient is assigned to the reactants and products after comparing the number of atoms of an element present on the reactant and product side. The stoichiometric coefficient of sodium carbonate, water, and carbon dioxide is 1 while that of sodium hydroxide is 2. Here is the reaction of chemical balancer: Then the stoichiometric coefficients are assigned to the molecules to describe the number of molecules of reactants and products participating in the reaction. The chemical equation balancer will provide the estimated complete balanced chemical equation.įirstly, the number of all atoms of each element on the reactant and the product sides are compared. Only enter the reactants if you are not given a complete unbalanced equation.The reactants and products are written in the form of symbols and formulae e.g., Co for cobalt. What is a Chemical Equation?Ī chemical equation is a symbolic representation of how reactants are converted to products in a chemical reaction. 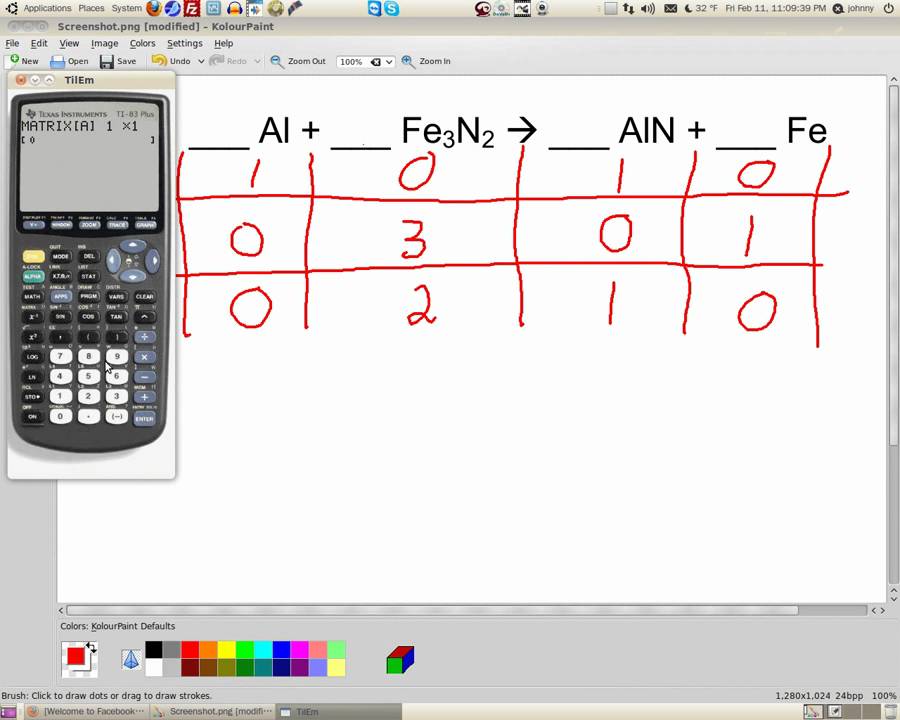
This article describes the process of balancing a chemical equation. One can use this online tool by following some simple instructions. The chemical equation balancer is here to help save time. Introduction to Chemical Equation Balancerīalancing a chemical equation is an important step while doing analytical calculations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
